
Dr. David Baron, PKD Foundation Chief Scientific Officer
Our very own Dr. David Baron shares his personal background with PKD, his journey joining the Foundation and his thoughts on the exciting news about the new treatment for ADPKD:
Little did I know when I was studying the effects of vasopressin and its modulators on toad urinary bladders at the University of Cambridge, that Dr. Jared Grantham, co-founder of the then PKR Foundation, had preceded me to Cambridge about a decade earlier doing seminal work on individual kidney tubules. It wasn’t until years later that I was diagnosed with PKD, the disease my father had (he died of a heart attack before his kidneys failed), my grandmother had and my great-grandfather must have had. A most generous kidney donor, with whom I had worked previously, Dr. Sandra Snook, flew to Chicago from San Diego for my transplant, just beating the arrival of a December 2009 snowstorm. Within two months, I was back at work. Sandra flew home a week after surgery! Within a year, I was “commuting” again to Osaka and Tokyo as Vice President for Nonclinical Safety Evaluation at a Japanese pharmaceutical company.
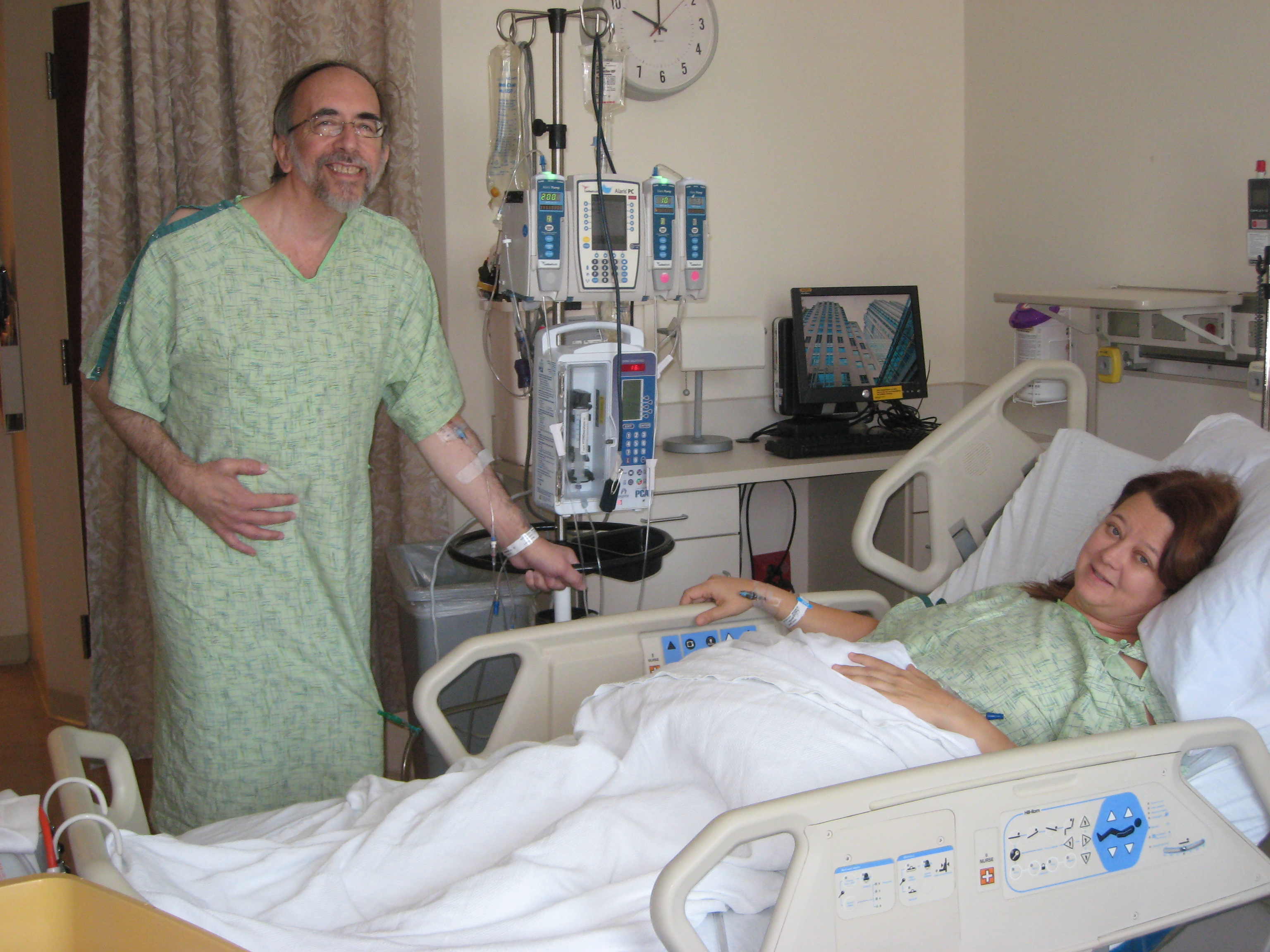
Dr. Baron and his kidney donor, Dr. Sandra Snook.
After a double nephrectomy (my PKD 30-pound diet), I retired, but having worked on pioglitazone, a type 2 diabetes drug, I had seen a few articles that showed that pioglitazone actually reduced the growth of cysts in rodent PKD models. The compound was nearing the end of its patent life, so my company didn’t want to test it in PKD patients, but I did. I looked for academic sites that I might persuade to conduct such a pilot study and came across the PKD Foundation website, which to my surprise was looking for a Chief Scientific Officer. I interviewed via Skype while on vacation in Jacksonville Beach, and after a long discussion with my wife and an intervening trip to Vietnam, Laos, and Cambodia, I decided to accept the offer even though it meant moving to Kansas City from Skokie, IL. The day after I returned from Vietnam, I flew to LA to attend my first PKD Foundation Board of Directors meeting. Sandra, once again thank you for sharing such a hearty kidney that lets me travel!
It’s been a privilege to have worked at the PKD Foundation the past 3+ years. I’ve met many of the clinicians and scientists (and clinician scientists) who are at the forefront of PKD research and translational medicine. It was also my distinct honor to have known Dr. Grantham – his scientific acumen, on which I often relied, was as sharp as it ever was even the week before he passed away.

Dr. Baron and Dr. Snook.
I’m happy to report that the Foundation, along with the U.S. Federal Drug Administration, is funding a pilot clinical study of pioglitazone in ADPKD patients in Indianapolis and even happier to report that tolvaptan is approved as the first drug in the U.S. for the specific treatment of ADPKD. The path through the regulatory minefield has been better defined thanks to the persistence of Otsuka Pharmaceuticals. I’m confident that the drug will open the door to future therapeutics for treatment of ADPKD and ARPKD. While it may be too late for me, as a transplant recipient, I am confident that the generation that follows will benefit from new therapeutics that will mitigate the worst effects of this devastating disease. With the promise of molecular genetics and breakthroughs like CRISPR technology – maybe we can entertain the possibility that a cure for PKD will eventually be found.









Going my to my nephrologist this week to see if I am in the running for Jynarque.
Very interesting. Thanks for posting this. Special thanks to Dr. Baron for his persistence.