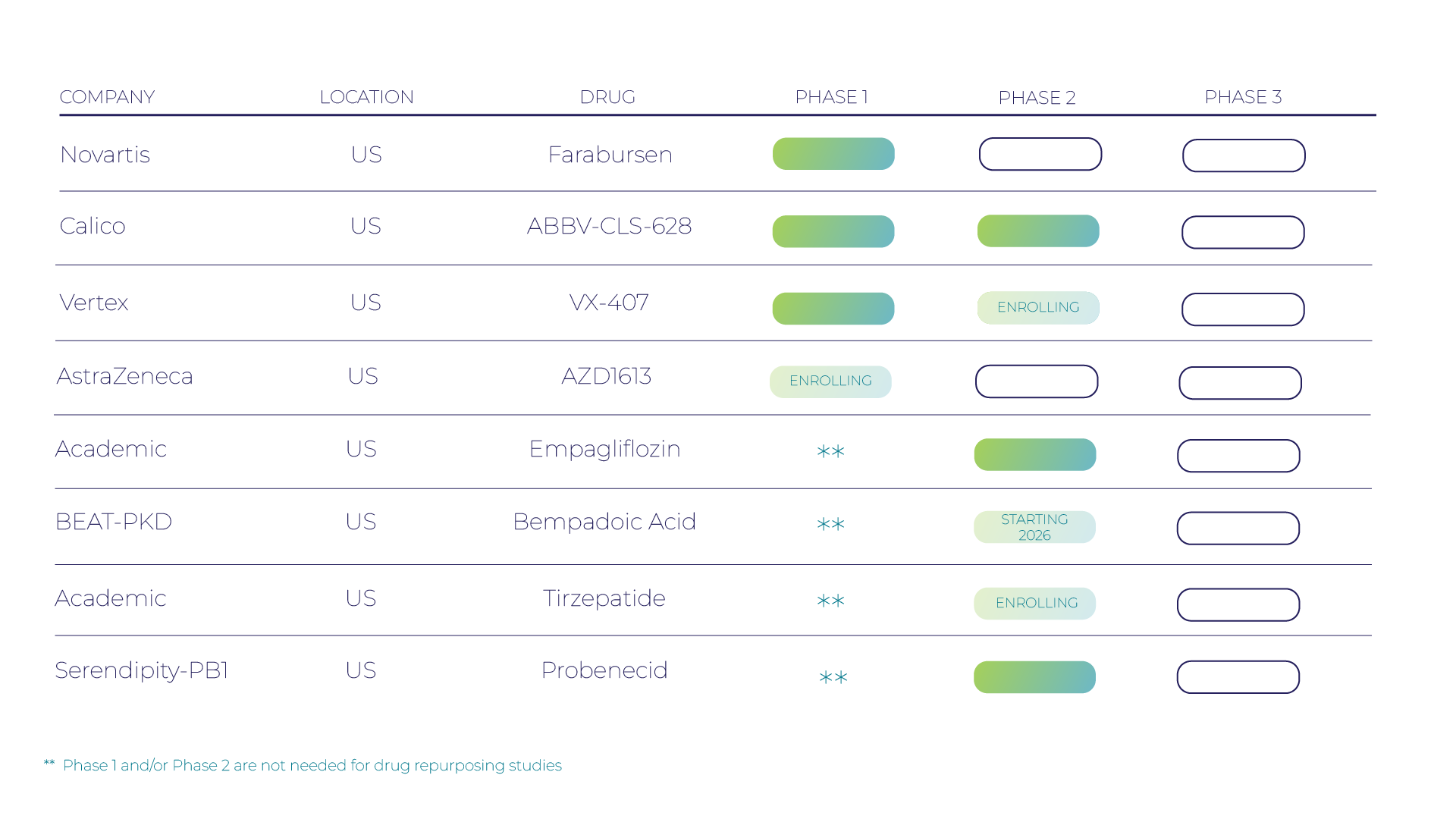
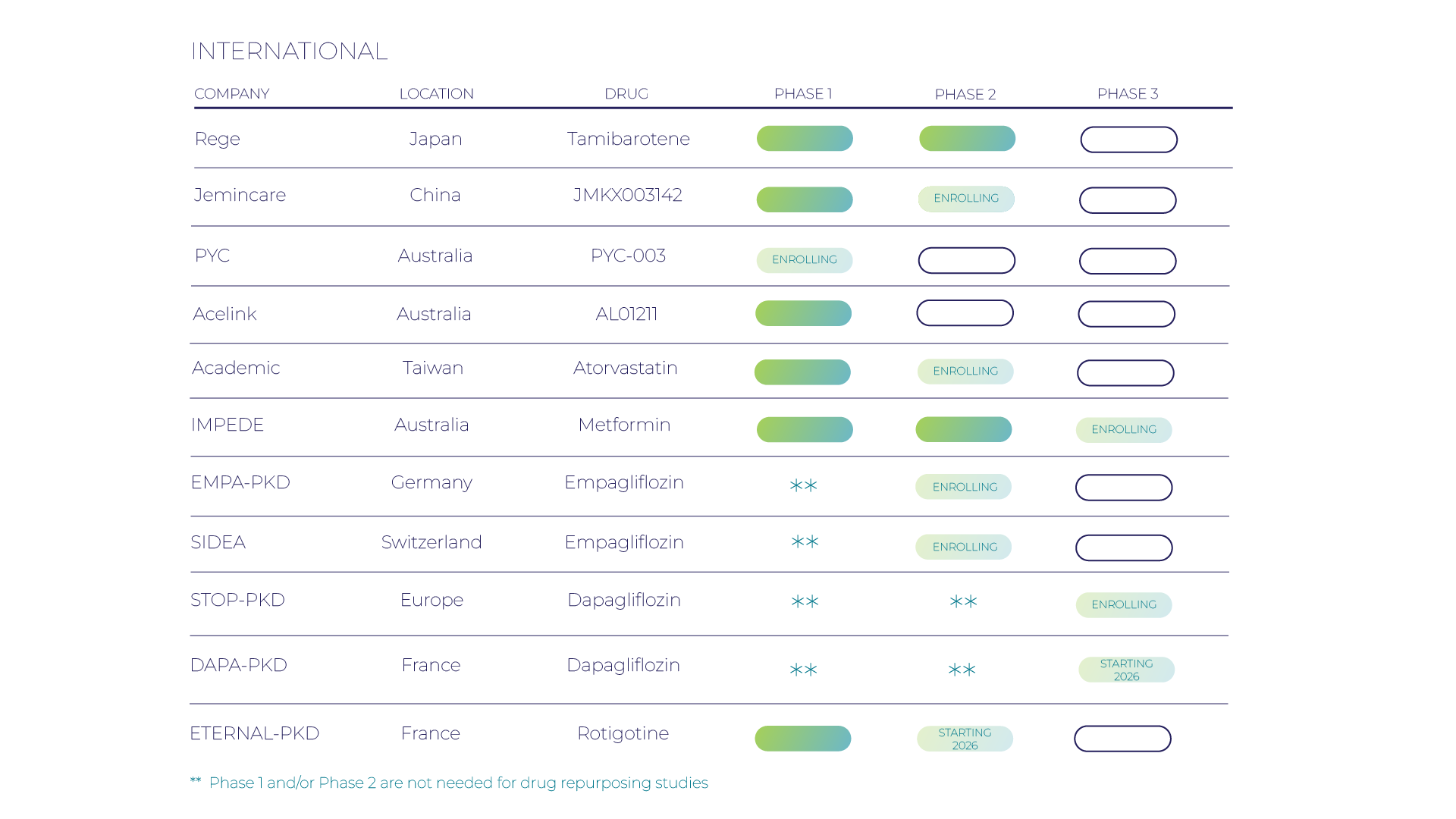
Research Pipeline
Since 1982, we’ve led the fight against PKD through the support of basic, translational, and clinical scientists; vital research funding; and patient education. Today, we’re encouraged by the significant strides we’re making to find treatments. We’ve gone from a single drug in clinical trials to an approved drug, tolvaptan, and more drugs in the pipeline today than ever before. Read on to learn more about the treatments for PKD currently being developed in the United States.


 Newly Diagnosed
Newly Diagnosed
A new diagnosis can leave you with many questions. Connect today to learn more about PKD and how we can help.
 Find a Community
Find a Community
Our local Communities, and the volunteers who lead them, are the backbone of the PKD community.
 Resources
Resources
With our resources you can find the answers to the questions you did and didn’t know you had about PKD.




